Abstract
- Superficial siderosis (SS) is a rare neurodegenerative condition characterized by hemosiderin deposition in the central nervous system, which sometimes leads to sensorineural hearing loss and vestibular dysfunction. This case report details the diagnosis and treatment of a 63-year-old female patient presenting with a yearlong history of postural instability and recent symptoms of sudden right-sided hearing loss and imbalance. Physical examination and imaging revealed bilateral deafness and infratentorial SS. Treatment, including medication, vestibular exercises, high-dose steroids, and hyperbaric oxygen therapy, led to a significant alleviation of dizziness but no improvement in hearing. This case emphasizes the need for clinicians to consider SS in patients presenting with sudden-onset hearing loss and dizziness, even without prior any medical history or trauma, to accurately identify the underlying cause.
-
Keywords: Superficial siderosis; Bilateral vestibulopathy; Hearing loss; Vertigo
-
중심단어: 표재철침착증, 전정장애, 청력 손실, 현훈
INTRODUCTION
Superficial siderosis (SS) is a rare neurodegenerative disorder characterized by the deposition of hemosiderin, an iron-containing pigment, in the superficial layers of the central nervous system, particularly the leptomeninges and subpial layers [1]. Although SS can be idiopathic, it may also result from bleeding associated with tumors, trauma, or arteriovenous malformations. The underlying pathogenesis is initiated by the destruction of red blood cells during hemorrhage, leading to iron deposition in glial cells and resulting in oxidation and cytotoxicity [2]. Although SS primarily affects the central nervous system, causing neurological symptoms, such as sensory and motor abnormalities and dementia, it may also induce sensorineural hearing loss and vestibular dysfunction [2]. Currently, the diagnosis of SS relies mainly on imaging techniques, including magnetic resonance imaging (MRI) [3]. To the best of our knowledge, only six cases have been reported in the literature describing the association between SS and bilateral vestibulopathy, as reported by Lee et al. [4], and two cases presenting with peripheral dizziness, as described by Kim et al. [3]. These patients exhibited varied clinical presentations. Recently, we encountered a case of bilateral sensorineural hearing loss and vestibulopathy induced by SS on MRI during the diagnosis and treatment of a patient with unilateral sudden sensorineural hearing loss, and we report it here along with a review of the literature.
CASE REPORT
A 63-year-old female patient presented to the outpatient clinic with a year-long history of postural instability and recent symptoms of right-sided hearing loss and imbalance, which had impaired her ability to walk 4 days prior to the visit. History taking showed a gradual progression of left-sided hearing loss over 20 years, which was confirmed as profound hearing loss a decade ago. Her medical history was notable only for hypertension, with no history of diabetes or hyperlipidemia. Both tympanic membranes were normal on otoendoscopic examination. Videonystagmography revealed mild rightward horizontal nystagmus at approximately 4°/sec, evident in spontaneous, gaze-evoked, and positional nystagmus. No increase in intensity was detected during the post-headshaking nystagmus test. Neurological examinations, such as the heel-to-shin test and the fingerto-nose test, performed to identify potential central lesions, did not show any suggestion of cerebellar dysfunction such as limb ataxia, and both motor and sensation were normal. In addition, no saccade or pursuit has been identified to suggest central lesions.
Pure-tone audiometry conducted on the day of the visit demonstrated bilateral deafness, and speech audiometry indicated a word recognition score of 0% in both ears (Fig. 1A). The patients underwent vestibular function testing on the same day. Video head impulse testing revealed decreased gain in all six semicircular canals (Fig. 1B), and the caloric test revealed 100% canal paresis on the left side and 100% right-directional preponderance. The spontaneous nystagmus velocities corrected for the maximum slow-phase velocity of nystagmus at each temperature stimulus were: 0°/sec for left cold and warm stimulation, 3.0°/sec for right warm stimulation, and ‒2.0°/sec for right cold stimulation (Fig. 1C). Additionally, both cervical and extraocular vestibular evoked myogenic potentials displayed no response, and computerized dynamic posturography indicated worse-than-normal performance in conditions 1, 2, and 3, and falls in conditions 4, 5, and 6 (Fig. 1D). Blood tests performed on the first day of hospitalization showed that hemoglobin, platelets, activated partial thromboplastin time, and prothrombin time were normal, and there were no bleeding factors. An MRI of the brain and internal auditory canal, conducted to discern the central origin of sudden hearing loss, revealed evidence of SS in the cerebellum. Treatment commenced on the day of admission with medication and customized vestibular exercises for dizziness, high-dose steroids (dexamethasone, 0.15 mg/kg/day), intratympanic steroid injection (0.3–0.5 mL of 5-mg/mL dexamethasone phosphate disodium), and hyperbaric oxygen therapy for right-sided sudden sensorineural hearing loss. Intratympanic steroid injections and hyperbaric oxygen therapy were continued daily during hospitalization, leading to her discharge 5 days later with no hearing improvement but significant dizziness alleviation.
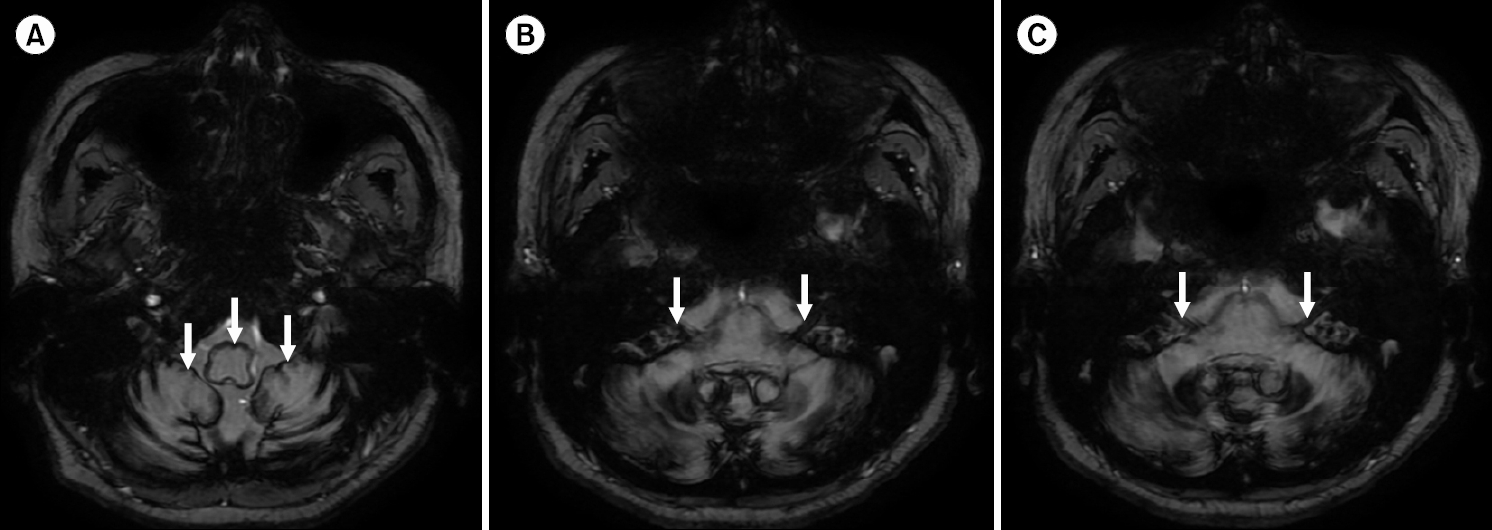
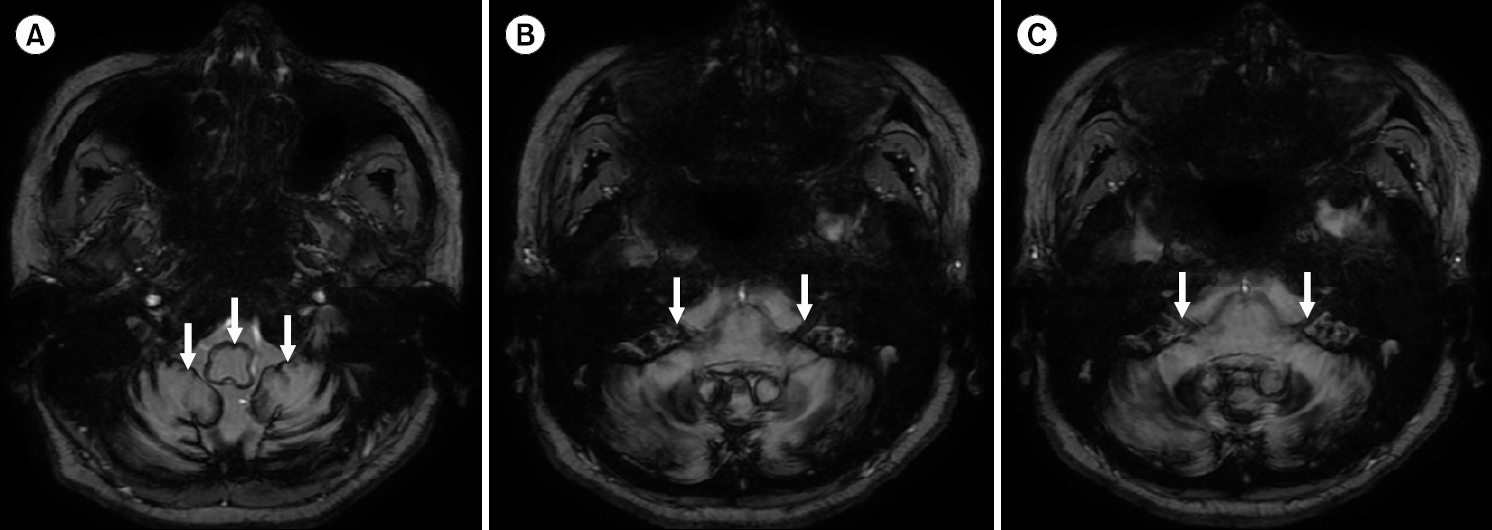
After discharge, the patient continued to receive high-dose oral steroids (prednisolone, 1 mg/kg/day) for 2 more days, followed by a 7-day taper, and an additional 26 days of hyperbaric oxygen therapy. Brain MRI was conducted for clear differentiation of a central cause, which exposed low-signal intensity SS throughout the cerebellum, involving the 7th and 8th cranial nerves (Fig. 2). After discontinuing steroids and completing 10 sessions of hyperbaric oxygen therapy, the patient reported reduced subjective dizziness (from 7 to 2 on the visual analog scale) and improved enough to walk independently on a 10-meter straight-line walk test. However, her hearing remained unchanged and a pure-tone audiogram confirmed bilateral deafness. She was then transferred to the neurology department of another hospital for further assessment and treatment of the SS.
DISCUSSION
Sudden deafness accompanied by dizziness can arise from various etiologies, including vascular diseases, viral inflammation, intralabyrinthine membrane rupture, autoimmune diseases, and tumors. Generally, these conditions are associated with a poorer prognosis than in patients without sudden deafness, thus necessitating an accurate diagnosis and immediate treatment [5]. Here, we describe a rare instance of sudden deafness coupled with vertigo caused by SS, which is a central lesion. This finding suggests that SS may also contribute to sudden deafness in patients with bilateral vestibular pathology, highlighting the complexity of such cases, even though they are common in clinical practice.
SS is an extremely rare neurodegenerative disease of the central nervous system that is poorly understood. It results from a hemorrhage into the subarachnoid space, often originating from various causes, such as intracranial tumors, spinal dural injuries, arteriovenous malformations, trauma, or metabolic diseases. Its pathogenesis involves damage to the dura mater by any of the aforementioned causes, leading to a hemorrhagic response and hemolysis. This process allows heme from red blood cells to invade the tissue, depositing iron (Fe) and causing deoxidation, cytotoxicity, cellular damage, and harm to glial cells, which can manifest as diverse neurological abnormalities depending on the affected area [2].
First described by Hamill [6] in 1908, SS was diagnosed through autopsy, which revealed iron deposits in cortical areas close to the cerebrospinal fluid in the 1940s [7]. The advent of MRI has shifted the diagnosis towards imaging examinations, mainly using T2-weighted susceptibility [8]. In the context of the cerebellum, a wider range of cortical folds and the long glial segment of the 8th cranial nerve increase the vulnerability to hematogenous invasion. Consequently, SS of the central nervous system typically leads to slowly progressive asymmetric sensorineural hearing loss and central or peripheral dizziness, primarily involving the cerebellum, 7th, and 8th cranial nerves. It may also cause dementia and sympathetic and parasympathetic disorders depending on the location and extent of iron deposits [2,4]. Although there was no central sign in our case, previous literature suggests that SS can cause both central and peripheral vestibulopathy [4]. In this specific case, MRI confirmed iron deposition in the cerebellum and along the 7th and 8th cranial nerves, leading to imbalance and bilateral sensorineural hearing loss without facial paralysis. SS is an irreversible condition resulting in iron-induced cytotoxicity and cell destruction. It is generally treated symptomatically when the source of the bleeding is unclear. Iron-chelating agents, such as deferiprone, a drug that crosses the blood-brain barrier, might be considered to alleviate symptoms and arrest disease progression. Previous studies have demonstrated reductions in hemosiderin levels and significant MRI changes in patients treated with deferiprone [9]. However, guidelines for administering iron-chelating agents remain undefined. In cases of profound bilateral hearing loss due to SS, such as the one presented here, the prognosis might be unfavorable because of the retrocochlear lesion. Cochlear implantation could be considered, as evidenced by reported improvements in sentence and monosyllabic word tests in some cases [4]. Recently, studies emphasizing early diagnosis and active surgical rehabilitation have reported average improvements in tone discrimination of >20% following cochlear implantation [10]. The patient in this case, who initially presented with sudden hearing loss and imbalance, received systemic high-dose steroid therapy, intratympanic steroid injections, and hyperbaric oxygen therapy. Despite these interventions, her hearing did not improve within 1 month of symptom onset. Therefore, swift consideration of cochlear implantation may be crucial for enhancing hearing capabilities.
Various forms of dizziness have been reported in eight previously documented cases of vestibulopathy caused by SS in Korea [3,4]. Among these, four cases with bilateral vestibular function abnormalities predominantly displayed combined central and peripheral patterns. Ocular motor tests in these cases revealed abnormal saccades and low pursuit gain, suggestive of a central lesion. Concurrently, these cases exhibited cerebellar ataxia and neuropathy, with bilateral caloric paresis representing a combination of central and peripheral vestibulopathy [4]. In contrast, our case lacked neurological indicators suggestive of central lesions, and no abnormal saccade or pursuit were discerned. Our patient did not experience stationary dizziness; however, a persistent imbalance that intensified in dim lighting conditions for over a year. According to other studies on combined peripheral and central vestibulopathy, central signals might be obscured by peripheral vestibular signals and vice versa [11,12]. Interestingly, in SS cases reported internationally, only three out of five cases displaying both central and peripheral vestibular damage confirmed cerebellar ataxia [8]. Therefore, in our case, while clinical presentations and vestibular function tests show peripheral vestibulopathy, the presence of an underlying central vestibulopathy cannot be dismissed. The bilateral maximal slow-phase velocity, measured at less than 6°/sec during the caloric test, along with a bilateral horizontal semicircular canal gain of <0.6 in the video head impulse test met the diagnostic criteria for bilateral vestibulopathy [13]. Although the patient initially presented with sudden unilateral hearing loss rather than progressive hearing loss, the primary vestibulopathy symptom was ataxia. After receiving combined medication and customized vestibular exercises, the patient’s ataxia significantly improved, and reports of dizziness in daily life became rare.
Earlier Korean cases of SS primarily involved patients with unilateral vestibulopathy with or without hearing loss, and bilateral vestibulopathy with progressive unilateral or bilateral hearing loss, often accompanied by a history suggestive of hemorrhage [3,4]. However, this case stands out as the patient's main complaint was sudden-onset unilateral hearing loss with imbalance, without any history of trauma or surgery that could lead to SS. The absence of these factors makes it challenging to suspect SS prior to MRI. Furthermore, trauma, autoimmune inner ear disease, meningitis, and ototoxic drugs are generally recognized as potential causes of bilateral loss of cochleovestibular function [14]. Hence, when a patient without a prior medical history presents with sudden-onset hearing loss coupled with dizziness and loss of cochleovestibular function, as in this case, careful evaluation is vital. Clinicians should scrutinize not only vascular disorders, autoimmune diseases, and tumors, but also the internal auditory canal and cerebellar lesions stemming from SS to accurately differentiate the underlying cause.
ARTICLE INFORMATION
-
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
FUNDING/SUPPORT
None.
-
AUTHOR CONTRIBUTIONS
Conceptualization, Formal analysis, Methodology, Project administration, Resources, Software, Visualization: GL, DBS; Data curation, Investigation: GL, YM; Supervision, Validation: DBS; Writing–Original Draft: GL, DBS; Writing–Review & Editing: All authors.
All authors read and approved the final manuscript.
Fig. 1.Results of audiometry and vestibular function test. (A) Pure-tone audiometry results show bilateral profound sensorineural hearing loss at the first visit (red, right ear; blue, left ear). (B) Result of vestibular function test performed on the first day after hospitalization. Video head impulse tests show the decrease of gain in both all semicircular canals. (C) Bithermal caloric test reveals canal paresis (CP) of 100% in the left ear and direction preponderance (DP) of 100% in the right ear on the second day of admission. (D) Sensory organization test results show conditions 1, 2, and 3, score below normal category and conditions 4, 5, and 6, a fall was observed at the second day of admission.
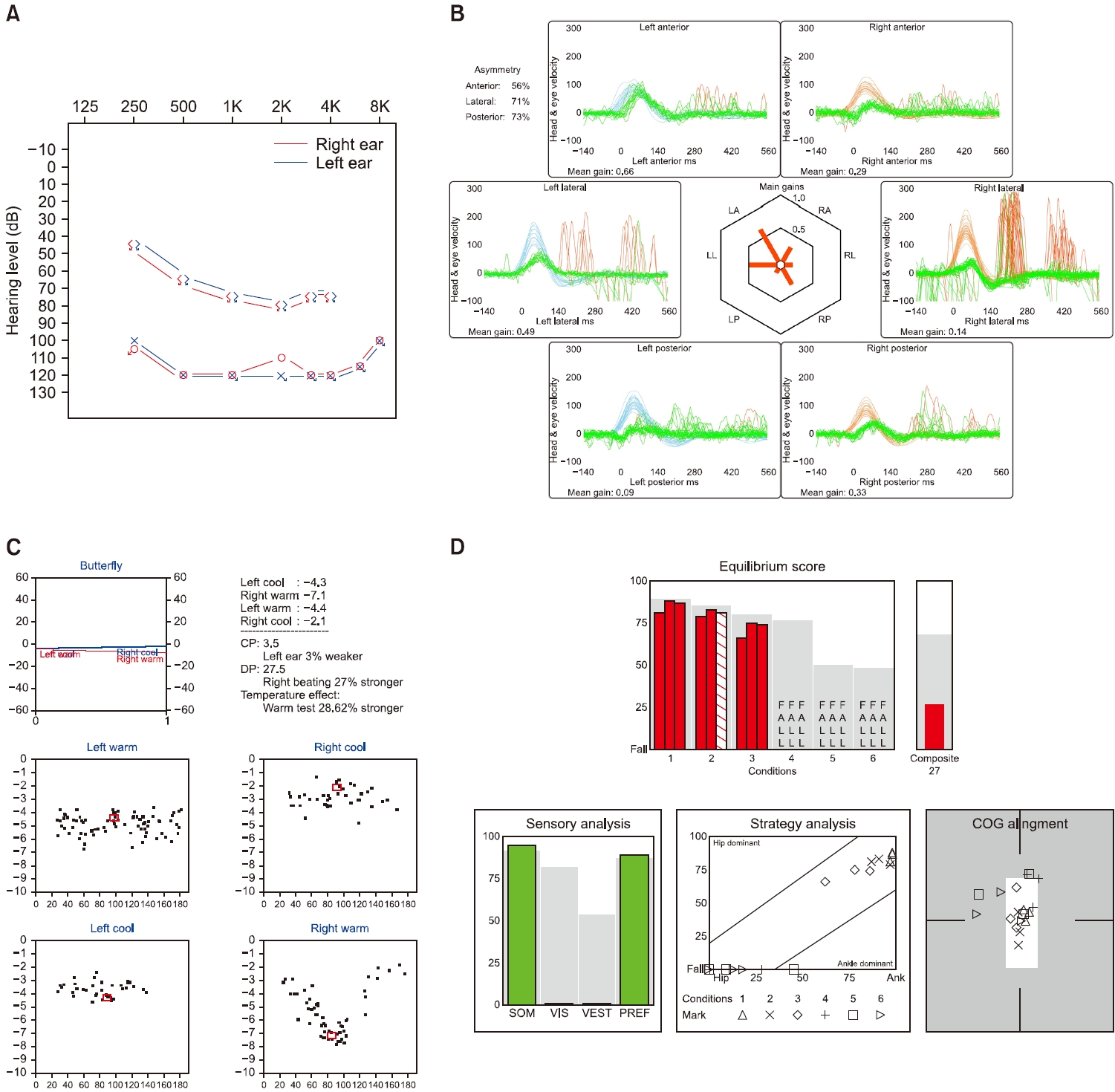
Fig. 2.Axial T2-weighted magnetic resonance images show (A) low-signal intensity, consistent with hemosiderin deposition, around cerebellum and brain stem (arrows), and (B,C) along the both 8th cranial nerves (arrows), and along the both 7th cranial nerve (arrows).
REFERENCES
- 1. Kumar N. Neuroimaging in superficial siderosis: an in-depth look. AJNR Am J Neuroradiol 2010;31:5–14.ArticlePubMedPMC
- 2. Kumar N. Superficial siderosis: a clinical review. Ann Neurol 2021;89:1068–79.ArticlePubMedPDF
- 3. Kim TH, Huh JH, Park MS, Byun JY. Superficial siderosis with peripheral dizziness: report of 2 cases. Res Vestib Sci 2018;17:60–6.ArticlePDF
- 4. Lee SY, Lee DH, Bae YJ, Song JJ, Kim JS, Koo JW. Bilateral vestibulopathy in superficial siderosis. Front Neurol 2018;9:422. ArticlePubMedPMC
- 5. Yu H, Li H. Association of vertigo with hearing outcomes in patients with sudden sensorineural hearing loss: a systematic review and meta-analysis. JAMA Otolaryngol Head Neck Surg 2018;144:677–83.ArticlePubMedPMC
- 6. Hamill RC. Report of a case of melanosis of the brain, cord and meninges. J Nerv Ment Dis 1908;35:594. Article
- 7. Noetzel H. [Diffusion von blutfarbstoff in der inneren randzone und äußeren oberfläche des zentralnervensystems bei subarachnoidaler blutung]. Archiv f. Psychiatrie 1940;111:129. –38. Germany.ArticlePDF
- 8. Miwa T, Minoda R, Matsuyoshi H. Vestibular function in superficial siderosis. BMC Ear Nose Throat Disord 2013;13:5. ArticlePubMedPMCPDF
- 9. Fredenburg AM, Sethi RK, Allen DD, Yokel RA. The pharmacokinetics and blood-brain barrier permeation of the chelators 1,2 dimethly-, 1,2 diethyl-, and 1-[ethan-1'ol]-2-methyl-3-hydroxypyridin-4-one in the rat. Toxicology 1996;108:191–9.ArticlePubMed
- 10. A rtukarslan E, Matin F, Donnerstag F, Gärtner L, Lenarz T, Lesinski-Schiedat A. Cochlea implantation in patients with superficial hemosiderosis. Eur Arch Otorhinolaryngol 2022;279:4363–70.ArticlePubMedPMCPDF
- 11. Choi SY, Kim HJ, Kim JS. Chasing dizzy chimera: diagnosis of combined peripheral and central vestibulopathy. J Neurol Sci 2016;371:69–78.ArticlePubMed
- 12. Choi KD, Lee H, Kim JS. Vertigo in brainstem and cerebellar strokes. Curr Opin Neurol 2013;26:90–5.ArticlePubMed
- 13. Strupp M, Kim JS, Murofushi T, Straumann D, Jen JC, Rosengren SM, et al. Bilateral vestibulopathy: diagnostic criteria consensus document of the Classification Committee of the Bárány Society. J Vestib Res 2017;27:177–89.PubMedPMC
- 14. Lee H, Yi HA, Baloh RW. Sudden bilateral simultaneous deafness with vertigo as a sole manifestation of vertebrobasilar insufficiency. J Neurol Neurosurg Psychiatry 2003;74:539–41.ArticlePubMedPMC
Citations
Citations to this article as recorded by







 KBS
KBS
 PubReader
PubReader ePub Link
ePub Link Cite
Cite